Drug Discovery and Lead Development Consultancy
Consultancy in natural products and chemical drug lead discovery using High Throughput Screening (HTS) technology, including their development into drug leads for clinical trials
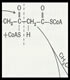
Screens of natural and synthetic chemicals have been the basis for the majority of groundbreaking leads that have opened up new frontiers in pharmacology in the past few decades. The provision of a robust assay, choice of hit criteria, artefact elimination, isolation and evaluation of the chemical hit, and assessing its potential as a lead, are all the activities covered by my high throughput screening (HTS) consultancy. The key issues here are how to provide a diverse chemical 'landscape', how to incorporate and source natural products, the provision of a low artefact assay and its automation, and how to recognise hits versus artefacts.
It is easy to get very large numbers of hits from the screening process but the next vital step is to turn a hit into a lead. This involves working with chemists and pharmacologists to identify any real leads present in the collection of hits. Obtaining and testing of analogues allows a structure activity relationship to be obtained. Target selectivity, knowledge of toxicology profiles of the chemical class and providing in-vitro toxicity testing can help to prioritise the hits. However, by far the best method of selecting a lead as well as validating the target is to show some in-vivo activity against a disease model. A disease model is also very useful for later stages of medicinal chemistry where the focus is often on bioavailability and toxicity. My consultancy will work with you to consider all these elements to allow lead progression up to Phase I clinical trials.
My Experience
In the late 1970s, working at Glaxo, I was involved with the development of some of the first high-throughput mechanistic screens that used isolated proteins as targets rather than whole cells or organisms. My bioprocessing skills in cell-culture, protein isolation and biochemical assay development were the reasons for my first entry into this field. After spending the next decade working in bioprocessing, in the 1990s I used my earlier experience of screening to propose, build and direct an entire HTS program at Monsanto-Searle to support new drug discovery. This involved the supply of millions of synthetic chemicals from libraries and combinatorial sources, and of natural product chemicals from plants, microbes, and marine sources.
Lead Discovery and Development Expertise:
- Technical and business criteria for selecting a drug target, including lead development considerations
- Designing and evaluating the HTS assay for a chosen target
- Requisite use of natural products, and potential benefits of using ethno botanical data and functional (cell based and whole animal) screening assays
- Evaluating libraries, and interfacing clients with suppliers of chemical and natural products libraries
- Integration of bio analytical methods with functional assays to decrease medicinal chemistry lead development timelines to Phase I clinical trials
- Assisting clients to find suitable partners for out-licensing drug leads and bio-analytical technology
- Overview of programs to decrease costs and improve throughput for clients using screening programs for both healthcare and agricultural lead chemical discovery
Contact me now to discuss how my Lead Discovery and Development consultancy can be used to improve your drug discovery programs.
Case Studies
Drug Discovery and Natural Products
A senior director of a US-based drug discovery company wanted me to assess the value of their natural product-screening program. I was able to provide the management with assurance that the team were technically highly competent with a wide diversity of genetic resources, thoughtful sample preparation and excellent structure elucidation technology. The issue for management was the extent to which natural products still had value compared with chemical library and fragment screening ethnology. I was able to provide them with convincing evidence that natural products were still providing cutting edge leads but that these properties were best realised if functional (cell or animal based) rather than purely mechanistic screens were used.
Agrochemicals Discovery
The discovery director of a US-based agricultural company wanted to me to review their screening technology. Over a number of visits I was able to build a high level of confidence with his staff and assist them in improving the management and integration of their program. I was also able to make a number of useful technical suggestions, including the use of quantitative imaging technology. In addition I provided them access to both chemical and natural product libraries for screening.
Improving the Diversity of Chemistry
The managing director of a UK company providing purified natural products pharmaceutical screens wanted me to assess their production and isolation technology. I was able to confirm that the products were of a suitable quantity and quality for high-throughput screening technology and but that their diversity of structural classes did not reflect the range of biological sources. I then suggested some modifications to their extraction procedures and as a result, a greater diversity of chemicals was identified.