Case Studies
- Bio-Business Development
- Manufacturing Biotherapeutics
- Manufacturing Natural Products and Biochemicals
- Drug Discovery and Development including high throughput screening
- Bioanalytical Technology and Markets
Out licensing of an Anti-fungal
A major US agricultural company, when screening for novel pesticides, had discovered a powerful anti-fungal agent. They were interested in whether it would have any value as a human anti-fungal medicine. Using my network I was able to obtain preliminary data showing a powerful anti-fungal activity against a spectrum of human pathogens. This became part of the data package which was sent to a select list of pharmaceutical companies whom I had identified as being potentially interested in such a lead chemical. The ability to raise such interest in an early stage lead was based on my ability to identify and target those companies that had active anti-fungal discovery programs and then target the discovery directors as well as the corporate in-licensing groups. The compound was successfully out-licensed after an extremely competitive bidding stage.
Market for Vacuum Dryer
A small UK manufacturer of drying equipment wanted me to tell them how their products were being used by the pharmaceutical industry. After giving the MD and senior staff a briefing about the role of their equipment in drug discovery programs, I was able to provide estimates of the market size and potential for future growth. This helped them to decide the extent to which they would expand their manufacturing facilities.
Market for Improved Biotherapeutic Proteins
A UK university spin-out company had a novel technology for a polymer that would improve the bioavailability of therapeutic proteins. They commissioned me to survey the existing market to see if there was any potential for them to use their technology to produce non-proprietary therapeutic proteins but with improved pharmacology. As a result, I was able to point to a small but significant market opportunity for their product. I was also able to identify potential partners and contract research organisations (CROs) that could help them progress down this avenue.
Acquiring an Analytical Company
A senior director in a highly successful analytical company, providing services to the water industry, was considering ways to expand the business. To help them survey the possibilities they asked me to identify any UK-based companies, providing services to the pharmaceutical industry that might be suitable for acquisition. I was able to provide a primer in the analytical support provided for pre-clinical and clinical research, potential for growth, and a short list of potential candidates. My consultancy helped them to make important decisions concerning the best way in which to grow their business.
Manufacturing Biotherapeutics
cGMP Cell Culture Pilot Plant
A German start up company wanted to evaluate whether it was more cost effective to outsource cGMP production of a recombinant protein by mammalian cell culture, or to build an in-house facility. My task was to provide the engineering group with a general area layout, equipment, people and product flow, and air quality scheme for the manufacturing suite. This information allowed them to consider the capital cost of the facility. I then listed all the capital equipment for up-stream and downstream processing, consumables and labour cost required to set-up and run the facility. I was able to point out the need to take into account the additional costs for process development and analytical labs. As a result of this input, the client decided that despite the local cash incentive to build the unit, outsourcing was the best solution.
Bioprocessing Cost Model
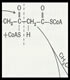
A UK university innovation company wanted me to evaluate the potential manufacturing cost of making a chemically modified peptide by rDNA technology. The peptide was previously made by peptide chemistry but this route was proving too expensive. I proposed making the peptide as a protein fusion in E.coli as the most effective route. We then built a comprehensive cost model based on the client’s estimated initial market need. An enzyme technology solution was proposed for the chemical derivatisation step which was shown to cost less than 30% of the chemistry route. I am now helping the client find a suitable contract research organisation to carry out the process development.
Bio-Pharmaceutical Project Planning
A mid-sized Danish pharmaceutical company, with little experience of therapeutic protein manufacture, wanted me to assess their project plan for making such a protein. Through my experience in fast-tracking such process development steps, I was able to advise them on all the parallel activities that would be required to pursue such a project. I also advised them to outsource the project to an experienced contract research organisation (CRO). This advice helped the client to create an achievable project plan and timeline for the project.
Manufacturing Natural Products and Biochemicals
Plant Cell Culture for Natural Products
An agricultural company based in Belgium wanted me to assess if a plant cell culture could be used to produce pharmaceutically-important small molecule natural products. My extensive knowledge of bio-manufacturing and the key role played by natural products as medicines meant that I was quickly able to review the potential of this technology. As a result I was able to advise them that this technology would be of very limited use.
Uses of Genetic Resources
A UK charity connected with Kew Gardens wanted to produce a map of all the uses that industry makes of genetic resources. Their initial focus was on the use of natural products in the pharmaceutical industry, so my role in natural products drug discovery programs put me in a good position to offer them advice. Furthermore, with my broad background in bioprocessing and biotechnology, I was able to expand their vision to include natural products used as pesticides for agriculture, biocatalysts for fine chemical manufacture, and the wide range of bio-indicators exploited by the analytical industry. I was also able to indicate the importance of these resources for making genetically modified organisms, and that these would be of use not only for food, but also for fibre and fuel production.
Plant Derived Nutraceutical
A Belgian university spin-out company wished to produce a plant-derived biopolymer that showed potential for the stimulation of ‘good’ gut bacteria. The biopolymer also showed potential for the treatment of inflammatory bowl disease. My task was to provide an evaluation of the product specifications, the current regulatory requirements needed to provide claims for efficacy, and the cost of clinical trials to support the nutraceutical vs. drug label claims. I was greatly helped in this task by the use of my network of regulatory and clinical trials contract research organisations. The results of my consultancy were integrated into the business plan for use in acquiring the next stage of funding.
Drug Discovery and Development
Drug Discovery and Natural Products
A senior director of a US-based drug discovery company wanted me to assess the value of their natural product-screening program. I was able to provide the management with assurance that the team were technically highly competent with a wide diversity of genetic resources, thoughtful sample preparation and excellent structure elucidation technology. The issue for management was the extent to which natural products still had value compared with chemical library and fragment screening ethnology. I was able to provide them with convincing evidence that natural products were still providing cutting edge leads but that these properties were best realised if functional (cell or animal based) rather than purely mechanistic screens were used.
Agrochemicals Discovery
The discovery director of a US-based agricultural company wanted to me to review their screening technology. Over a number of visits I was able to build a high level of confidence with his staff and assist them in improving the management and integration of their program. I was also able to make a number of useful technical suggestions, including the use of quantitative imaging technology. In addition I provided them access to both chemical and natural product libraries for screening.
Improving the Diversity of Chemistry
The managing director of a UK company providing purified natural products pharmaceutical screens wanted me to assess their production and isolation technology. I was able to confirm that the products were of a suitable quantity and quality for high-throughput screening technology and but that their diversity of structural classes did not reflect the range of biological sources. I then suggested some modifications to their extraction procedures and as a result, a greater diversity of chemicals was identified.Bioanalytical Technology and Markets
Uses of Analytical Technology
A UK-based instrument development company wanted to know if there was any potential for their proprietary analytical technology to be used for high throughput screening applications. I told them that its high sample cost and low throughput meant that it would have very little application in that area. However, I felt that it would have considerable value for the diagnostics business and was able to put them in contact with key players in this market.
Analytical-Specifications
A bio-pharmaceutical discovery company had outsourced the mammalian cell production of their lead protein. This work was going well, but they were concerned about the regulatory and quality control aspects of a PEGylation step. With my experience in GMP manufacturing and specification setting, I was able to suggest the sort of pre- and post-PEGlyation specifications required. I also advised them on the GMP requirements for the chemical derivatisation and discussed the in-process controls that might be established. This helped to reassure them that such a product could be made to acceptable quality standards.
Dispensing Technology
A UK-based analytical instrument company had acquired a micro-droplet dispensing technology. They wanted to know if there was a market for this in the pharmaceutical industry. I was able to brief them on the current state of analytical technology used in both R&D and diagnostics and point them to the most likely applications of this technology. This helped them to decide which markets to target with their new technology.